SAH, Methylation, and Mood: A Functional Medicine Perspective"
Managing Elevated SAH: Effects and Interventions
Mood disorders are linked to biochemical changes, particularly concerning the levels of S-adenosylhomocysteine (SAH). An increase in SAH levels impacts methylation processes, especially in the DNMT histone methylation pathway. This can disrupt serotonin functions vital for mood regulation and lead to imbalances in neurotransmitters. The resultant effects can manifest as mood disorders, such as OCD, chronic depression, and schizoaffective disorder. These disorders are sometimes misdiagnosed because the underlying biochemical imbalance is overlooked.
SAH Elevation: Biochemical Pathways and Implications
Understanding SAH and Its Origins:
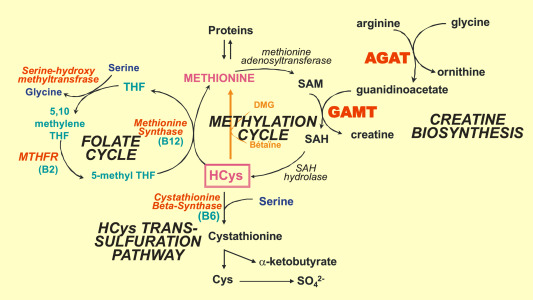
SAH, or S-adenosylhomocysteine, is a molecule central to methylation, a key cellular process. Its rise is intricately tied to SAMe (S-adenosylmethionine), a molecule that donates a methyl group during reactions facilitated by methyltransferases.
Significance of Methyltransferases:
Methyltransferases, enzymes central to the methylation process, handle the transfer of methyl groups from SAMe to various substrates. The performance and integrity of these enzymes are essential for several biochemical pathways:
Methyltransferases: Key Players in Cellular Processes
Methyltransferases are vital enzymes governing diverse cellular tasks:
- DNA Methyltransferases (DNMTs): DNMT histone methylation pathway impacts the activity serotonin and dopamine by increasing the expression of transporters SERT and DERT. Serotonin and dopamine vital neurotransmitters, and any disruption in its synthesis or transportation can lead to mood and behavior disorders.
- Catechol-O-Methyltransferase (COMT): Regulates the methylation and deactivation of neurotransmitters like dopamine, potentially affecting mood.
- Phosphatidylethanolamine N-Methyltransferase (PEMT): Essential for cellular health, transforming phosphatidylethanolamine to phosphatidylcholine in cell membranes.
Neurotransmitter Regulation and Genetic Mechanisms: An Overview
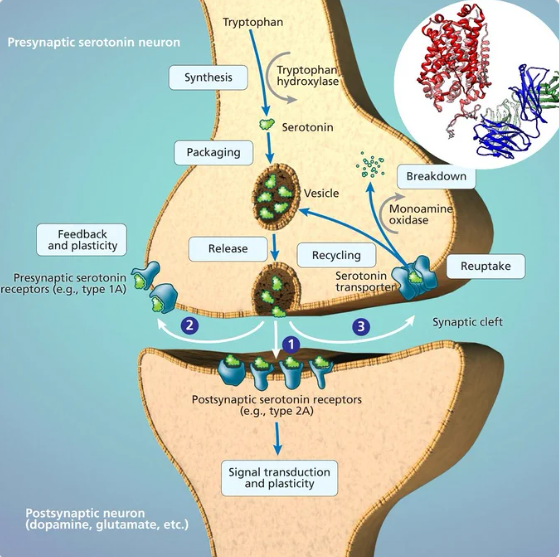
The brain, comprising a vast network of neurons, communicates using neurotransmitters. For efficient messaging, neurotransmitter activity must be tightly regulated. Both the serotonin and dopamine transporters, SERT and DAT respectively, serve to reclaim these neurotransmitters after their release. Their production and activity are controlled by specific genes, with complex genetic processes like DNA methylation and histone modifications modulating their levels and, consequently, the neurotransmitter signaling.
SERT (Serotonin Transporter):
Produced by serotonergic neurons primarily in the raphe nuclei of the brainstem, SERT reabsorbs serotonin from the synaptic cleft back into the neuron, terminating its signaling action.
Epigenetically, the SLC6A4 gene promoter, encoding SERT, can be influenced by DNA methylation changes. Histone modifications, notably H3K4me3 and H3K27me3, play crucial roles in SERT expression. Undermethylation in regions associated with SLC6A4 gene repression might elevate SERT expression, thereby reducing serotonin signaling.
DAT (Dopamine Transporter):
Dopaminergic neurons, mainly found in the substantia nigra and ventral tegmental area (VTA) of the midbrain, produce DAT. This transporter ensures dopamine reuptake from the synaptic cleft.
Like SERT, DAT expression can be epigenetically influenced. Activating marks like H3K4me3 tend to increase DAT expression, while repressive marks like H3K27me3 decrease it. Undermethylation in repressive DNA regions can amplify DAT expression, curtailing dopamine signaling due to enhanced reuptake.
The nuances of these genetic mechanisms operate within a dense web of cellular interactions. The influence of undermethylation on SERT and DAT is interwoven with numerous factors, such as the specific change locations and overall cellular milieu.
Increased SAH levels can disrupt these methyltransferases, leading to cellular disturbances and broader physiological challenges. Understanding these pathways and their interplay with SAH provides a foundation for deeper biological insights.
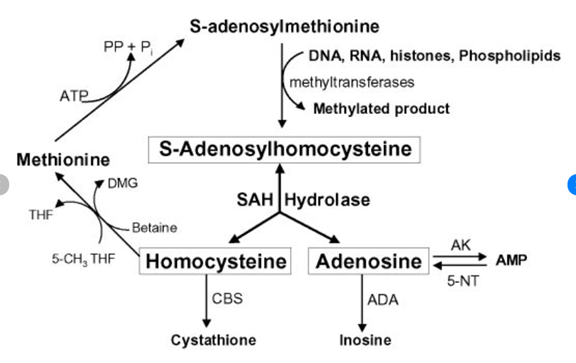
Introduction to SAH Metabolism and Its Importance in Liver Function:
S-Adenosylhomocysteine (SAH) metabolism holds a significant position in the biochemical equilibrium of the human body. The conversion process from SAH to homocysteine (HCY) is imperative for the effective functioning of the liver, primarily within the Phase II transsulfuration pathway. This vital detoxification route, when efficiently operational, permits the liver to have increased substrate HCY to further feed the transsulfuration pathway. The downstream result of this pathway is the synthesis of glutathione, a pivotal antioxidant for the body. Glutathione plays an indispensable role in neutralizing various toxins, easing the liver's burden and preventing potential accumulation of SAH, which can emerge due to an augmented demand for transmethylation, consuming notable ATP amounts. In this light, ATP's role becomes increasingly evident, as it aids both adenosine removal and the essential conversion of SAH to HCY.
The Dynamics of ATP and Its Role in Cellular Functionality:
ATP, scientifically termed Adenosine Triphosphate, is the cellular energy currency, acting as the primary energy facilitator for numerous cellular operations. The energy trapped within ATP is released during the cleavage of its high-energy phosphate bonds, which in turn, powers a plethora of cellular processes, including muscle contractions and the synthesis of neurotransmitters. The synthesis, degradation, and utilization rates of ATP are accurate indicators of a cell's metabolic vigor and overall health. Beyond its general cellular roles, ATP is instrumental in methylation cycles, where it ensures seamless conversions, notably that of SAH to HCY. For liver detoxification, the availability of ATP in abundant quantities means that the liver can operate at peak efficiency, processing toxins and ensuring that detrimental compounds are rapidly expelled, preserving the biochemical homeostasis.
S-adenosylhomocysteine (SAH) and Methylation Dynamics:
Perturbations in SAH levels in the human body can be attributed to two prevailing mechanisms. Firstly, an excess of adenosine can stymie the efficiency of the enzyme SAHH, leading to an accumulation of SAH as the conversion process to homocysteine is hampered.
Zinc's Role in Biochemical Pathways:
Zinc, an essential trace mineral, is pivotal for a myriad of physiological processes, notably the conversion of SAH to HCY by the enzyme SAHH. Its role as a necessary cofactor ensures the robust and efficient functioning of this enzyme. Further, zinc's role extends to the spheres of cellular energy dynamics, underpinning mitochondrial function and ensuring efficient ATP production. Zinc provides structural support to mitochondrial membranes, fortifying them against oxidative stress. Moreover, in the detoxification spectrum, zinc plays dual roles: acting as a cofactor for specific cytochrome P450 enzymes and aiding in the synthesis of metallothioneins, proteins essential for sequestering heavy metals. When zinc is coupled with antioxidants, its efficacy in defense mechanisms against oxidative stress is magnified, reinforcing mitochondrial function and expediting the detox process.
Potential Therapeutic Regimens for Increasing ATP Production:
- Methylene Blue: Enhances mitochondrial respiration and ATP production.
- Magnesium: Essential for ATP synthesis.
- Coenzyme Q10 (CoQ10): Converts nutrients into ATP and acts as an antioxidant.
- L-Carnitine: Transports fatty acids into mitochondria, boosting energy production.
- Creatine: Involved in ATP formation.
- Alpha-lipoic acid (ALA): Part of mitochondrial energy pathways and offers antioxidant properties.
- Adenosine Monophosphate (AMP): Indicates cellular energy status.
- Ozone Therapy: Boosts oxygen levels, enhancing ATP production.
- NAD (Nicotinamide Adenine Dinucleotide): Facilitates redox reactions, aiding energy conversion.
- Zinc in ATP Production and Mitochondrial Health: Essential for mitochondrial function and ATP production.
Liver Detoxification and Transsulfuration Pathway
Significance of Liver Function in SAH and Adenosine Metabolism:
The liver stands as a pivotal organ for numerous metabolic pathways, most notably regarding methionine metabolism, the clearance of homocysteine, and the metabolism of both SAH and adenosine.
Role of the Liver in Methylation: Central to detoxification processes, the liver utilizes methylation to neutralize and expel a variety of substances, which include toxins and medications. This vital process prominently features S-adenosylmethionine (SAMe). Following its methyl group donation, SAMe transitions into S-adenosylhomocysteine (SAH). However, complications like non-alcoholic fatty liver disease (NAFLD) can impede these methylation operations. Such conditions may force the liver to use more methyl groups for processing fats and toxins, leading to an increased conversion of SAMe to SAH. Furthermore, liver damage can impede the regeneration of SAMe, potentially disturbing the SAMe to SAH equilibrium. The story grows more intricate with obesity, as it can exacerbate liver functionality, especially if it induces NAFLD. Chronic inflammation from obesity, marked by a surge in inflammatory cytokines, amplifies demands on the liver's methylation processes. Complicating matters further, insulin resistance, a frequent companion of obesity, can alter essential substrates for methylation due to shifts in insulin sensitivity and glucose metabolism.
In the tapestry of liver function, phosphatidylcholine emerges as a vital player, aiding in the transport of fat and cholesterol within the liver. A dearth of phosphatidylcholine could cause the liver to increase its consumption of SAMe to compensate, culminating in elevated SAH levels. For SAH, the subsequent metabolic phase is its conversion to homocysteine. However, certain deficiencies, like that of vitamin B, can halt this transition, leading to an accumulation of SAH. Some medications, such as proton pump inhibitors, may reduce the absorption of essential nutrients like vitamin B12, thereby complicating SAH's conversion.
Therapeutic Approaches and Recommendations:
Reducing Toxin Exposure: Minimizing contact with harmful agents—like alcohol, certain drugs, environmental toxins, and food contaminants—can lighten the liver's load.
Liver Support: Employing antioxidants, colchicine, dandelion, N-acetyl cysteine (NAC), lipase, and digestive enzymes can be beneficial. Additionally, detoxification methods like the Hulda Clark gallbladder flush and coffee enemas deserve mention.
Dietary and Gut Health: Prioritizing a nutrient-rich and minimally processed diet is vital for liver health. Also, nurturing a balanced gut flora can fortify liver functions given the intimate relationship between liver and gut processes.
The Liver, Glutathione Production, and Toxin Removal:
The liver, our body's primary detoxification organ, intricately connects to the processes of methylation and adenosine metabolism. At the heart of this nexus is homocysteine (HCY), a vital intermediate in the methylation pathway. The liver employs HCY in the production of glutathione, a master antioxidant and detoxifying agent, via the phase 2 transsulfuration pathway. Adequate levels of glutathione are imperative for our liver to effectively neutralize and remove toxins, including heavy metals. This process, termed phase 1 detoxification, is facilitated by specific enzymes which can be enhanced with supplements and nutrients.
Several compounds have emerged as vital supporters of these liver processes. L-Serine, for instance, plays a pivotal role in glutathione synthesis, while N-acetyl cysteine (NAC) directly replenishes glutathione stores. Vitamin B6 acts as a coenzyme in the transsulfuration pathway, assisting in the conversion of HCY to cysteine, a precursor for glutathione. As these pathways are bolstered, the liver becomes more efficient in toxin removal, subsequently reducing the likelihood of SAH accumulation and its inhibition on methylation.
Yet, our liver is constantly besieged by multiple challenges: the accumulation of fats leading to fatty liver disease, regular ingestion of processed foods, environmental toxins, and even gut-derived endotoxins due to impaired intestinal health. Each of these factors pulls on the liver's glutathione reserves, potentially leading to a deficit. By minimizing these burdens, we not only preserve glutathione but also ensure that cells are energetically optimized, with ATP production being balanced.
A Holistic Outlook on Health and Methylation:
In essence, the pathways of methylation, ATP production, and liver detoxification are tightly interwoven. By ensuring efficient conversion of SAH to HCY, we empower the liver to synthesize more glutathione. This not only amplifies our detoxification prowess but also redirects HCY towards the formation of methionine and subsequently, S-adenosylmethionine (SAMe). SAMe is a universal methyl donor, critical for numerous methylation reactions that are paramount for our health. Thus, a well-functioning liver, efficient methylation pathways, and balanced ATP production collectively synergize, offering us the foundation for optimal health.
Liver Health and Toxin Elimination
- Minerals for Liver Health:
- Iron: Supports oxygen transport.
- Copper: Involved in oxidative phosphorylation.
- Sulfur: Enhances cellular respiration.
- Improving Phase I and Phase II detoxification: Consider compounds like sulforaphane (from cruciferous vegetables), N-acetylcysteine, and milk thistle.
- Phosphatidylcholine: Beneficial for liver cell membrane health and supports bile flow.
- Gall Bladder Health: Regular intake of healthy fats, beetroot, and bitter greens can stimulate bile flow. Taurine can also be considered to support bile acid conjugation.
- Zinc in Enzymatic Function and Methylation: Supports SAHH function, which is crucial for converting SAH to homocysteine.
- Dandelion (Taraxacum officinale): Historically, dandelion has been used to treat liver problems. The root is believed to act as a liver tonic by stimulating bile production and eliminating toxins. Dandelion may also have diuretic properties, which can help the liver quickly eliminate toxins.
- Artichoke (Cynara scolymus): Artichoke leaves are believed to stimulate bile production, which helps to metabolize fats in the liver and promote detoxification. Artichoke might also help protect liver cells from damage.
- Burdock (Arctium lappa): Traditionally used as a blood purifier, burdock root can help cleanse the blood of toxins. It's believed to stimulate bile flow and support overall liver function.
- Milk Thistle (Silybum marianum): Often taken for liver support, milk thistle contains silymarin, a compound known for its hepatoprotective properties. Silymarin not only helps shield liver cells from toxins but also supports liver regeneration and the elimination of harmful substances.
- Turmeric (Curcuma longa): Widely consumed for its potent anti-inflammatory and antioxidant properties, turmeric is rich in curcumin. Curcumin promotes liver health by combating inflammation, aiding in detoxification, and helping protect the liver from damage.
The Liver's Role in Detoxification and the Influences that Affect its Functionality:
The liver serves as the body's central detoxification hub, working tirelessly to neutralize harmful substances. Its efficiency, however, can be affected by specific foods, toxins, medications, and lifestyle choices. Being aware of these factors can assist in fostering liver health.
- Foods and Beverages Influencing Liver Health:
- Alcohol: Chronic and excessive intake is associated with liver diseases such as alcoholic hepatitis, fibrosis, cirrhosis, and fatty liver disease.
- High-Fructose Corn Syrup: Abundant in processed foods and sugary beverages, its overconsumption is linked to non-alcoholic fatty liver disease (NAFLD).
- Fatty Foods: Trans fats and certain saturated fats are known contributors to fatty liver conditions.
- Aflatoxins: Produced by specific molds, these mycotoxins can contaminate crops, including corn and peanuts. Continuous exposure raises liver cancer risks.
- Red and Processed Meats: Some research suggests a correlation between these meats and heightened liver disease risks.
- Medications Impacting Liver Functionality:
- Acetaminophen (Tylenol): It's imperative to adhere to recommended dosages. Overdose is a primary cause of acute liver failure in the U.S.
- Certain Antibiotics: Antibiotics like amoxicillin-clavulanate and isoniazid may induce liver damage in rare instances.
- Statins: While essential for cholesterol management, occasional use may result in liver damage.
- Non-prescription Pain Relievers: NSAIDs, for instance, can occasionally affect liver processes.
- Chemicals and Their Effects on the Liver:
- Pesticides and Herbicides: Exposure, whether occupational or via residues on non-organic produce, can stress the liver.
- Bisphenol A (BPA): Found in select plastics, BPA can disrupt liver function.
- Heavy Metals: Including lead, mercury, cadmium, and arsenic, these metals can come from sources like contaminated fish, old paint, and certain rice types.
- Perfluorinated Chemicals: Used in non-stick items and specific food packaging, these compounds can accumulate in the liver.
- Polychlorinated Biphenyls (PCBs) and Dioxins: Previously used in electrical setups and still detected in some seafood and meats, these toxins can damage the liver.
-
- Lifestyle Choices and Other External Factors:
- Tobacco Smoke: Smoking introduces numerous toxic compounds into the body, impacting liver health over time.
- Obesity: A leading contributor to NAFLD, it is a significant concern for liver health.
- Viral Infections: Viruses like Hepatitis B and C can trigger chronic liver ailments and even liver cancer.
- Herbal Supplements and Liver Health:
- Kava Kava: Although consumed for its soothing effects, it's been associated with potential liver damage.
- Ma Huang (Ephedra): This herb has links to liver damage in certain cases.
- Comfrey: Rich in pyrrolizidine alkaloids, this herb can be detrimental to the liver.
Digestive Health:
- Zinc: Supports gastric acid production and used in zinc-carnosine, beneficial for gut health.
- Therapies for Leaky Gut: Consider collagen peptides, bone broth, L-glutamine, and probiotics to repair intestinal lining.
- Intestinal Dysbiosis Therapies: Probiotics, prebiotics, and dietary changes to restore gut flora balance.
- Toxin Ingestion: Activated charcoal, bentonite clay, and zeolite can bind toxins for elimination.
- Zinc in Digestive Health: Essential for gastric acid production and aids in gut integrity.
Utilizing a functional medicine approach, the correlation between SAH, mood disorders, and methylation has been highlighted as essential in understanding mental health. Our dietary habits, environmental factors, lifestyle choices, and supplements all play a significant role in influencing this relationship. This comprehensive perspective emphasizes the importance of considering all these elements for optimal mental health, promoting a balance in these essential biochemical pathways.