Methylation and Mitochondrial Dysfunction in Autism
Autism is often discussed in terms of behavior or learning, but behind those challenges lies a hidden biological story—how the body produces and manages energy. Research on mitochondria in autism and methylation has revealed that many individuals on the spectrum share a pattern of low cellular energy, high oxidative stress, and difficulty maintaining biochemical balance.
Mitochondria are the body’s power plants, generating the energy (ATP) that keeps every organ, especially the brain, functioning. Methylation, meanwhile, controls how efficiently that energy is produced and protected. When methylation slows, the body struggles to make key molecules such as glutathione and creatine, which mitochondria depend on to stay healthy. This overlap between mitochondrial weakness and poor methylation explains much of the fatigue, mood instability, and sensory sensitivity often seen in autism.
What Mitochondria Do
Mitochondria are tiny power plants inside nearly every cell. Their main job is to turn nutrients—especially fats, glucose, and amino acids—into ATP, the molecule that fuels everything from brain signaling to muscle movement. When mitochondria work well, the body maintains stable energy, clear thinking, and calm mood regulation.
In mitochondrial dysfunction in autism, these energy-producing systems don’t run efficiently. The brain, which demands enormous amounts of ATP, can become energy-deprived. This contributes to symptoms like brain fog, poor focus, low stress tolerance, and fatigue. The same imbalance also affects digestion, immunity, and detoxification—systems that rely heavily on consistent cellular energy.
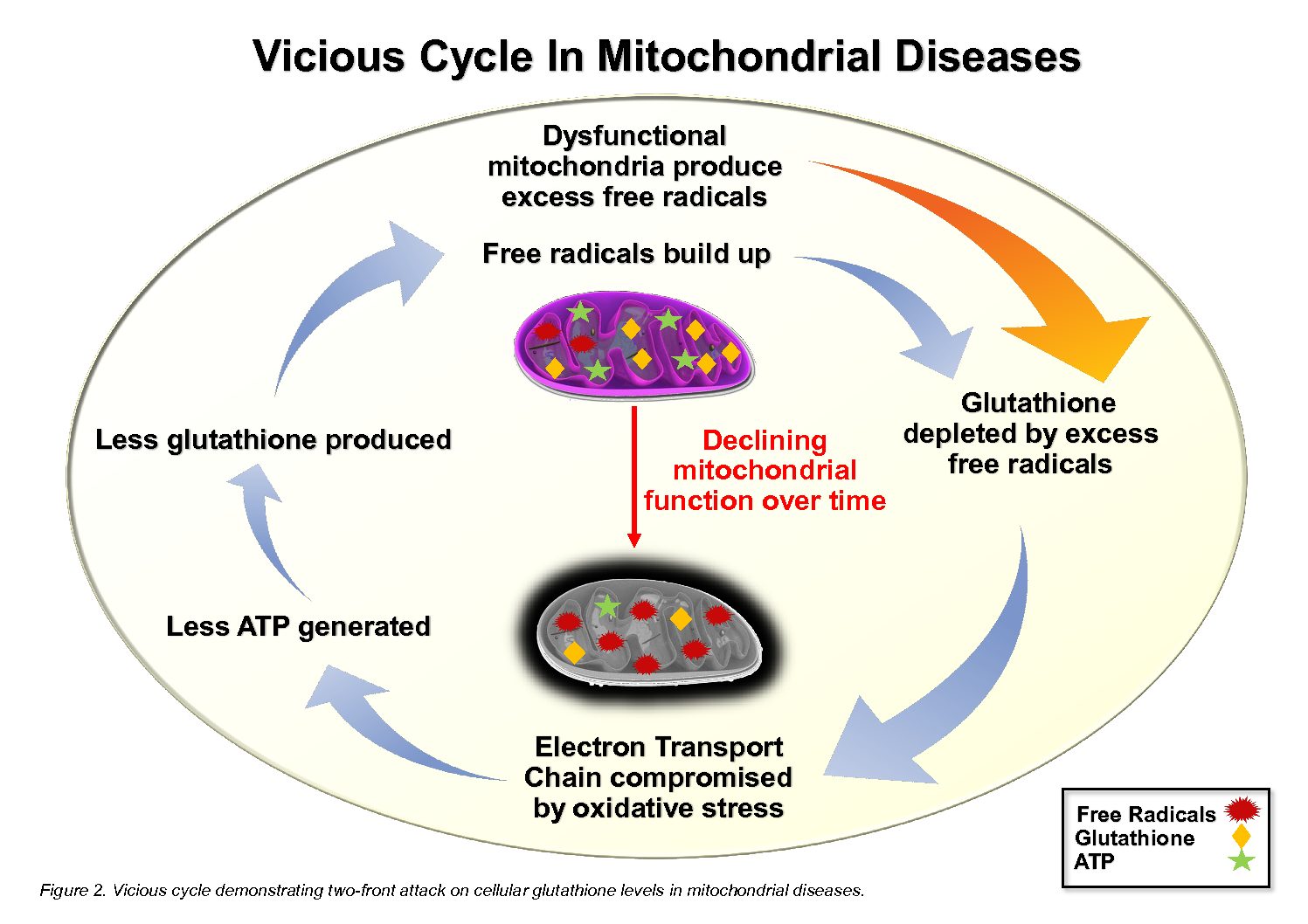
Mitochondria are also responsible for maintaining redox balance—managing the natural by-products of metabolism known as reactive oxygen species (ROS). When this process falters, oxidative stress rises, damaging enzymes and cell membranes. The body then needs strong antioxidant systems—like glutathione, catalase, and superoxide dismutase—to neutralize these molecules. Without enough methylation support, however, the body struggles to make glutathione, creating a cycle where energy production problems in autism feed oxidative stress, and oxidative stress further weakens mitochondrial function.
How Methylation Keeps Mitochondria Running
Methylation is one of the body’s most important biochemical systems — the process that donates small chemical groups (methyl groups) to regulate DNA, enzymes, and neurotransmitters. This mechanism controls energy production, detoxification, and emotional balance.
According to Dr. William Walsh’s research, up to 90% of individuals with autism are undermethylated. In this state, there is often low SAM (S-adenosylmethionine) and elevated SAH (S-adenosylhomocysteine), reflecting reduced methyl group availability. Because SAM is required for hundreds of methylation reactions, including those that build creatine and glutathione, low levels of SAM can ripple across nearly every body system.
- Creatine is vital for mitochondrial energy buffering — it stores and transfers phosphate groups that recycle ATP, the cell’s main energy currency. Without adequate SAM, creatine synthesis slows, leaving brain and muscle cells more vulnerable to fatigue and poor resilience under stress.
- Similarly, glutathione, the body’s master antioxidant, depends on both methylation and the transsulfuration pathway that follows it. When methylation falters, homocysteine and SAH accumulate, limiting the production of cysteine and glutathione. The liver and mitochondria then lose their main defense against oxidative damage, allowing inflammation and toxin buildup to worsen.
This is why Walsh’s approach emphasizes balancing methylation—not with folates, which can worsen symptoms in undermethylated individuals—but with nutrients that rebuild the methyl cycle naturally. These include zinc, magnesium, vitamin B6 or P-5-P, methionine, N-acetylcysteine (NAC), and creatine. Together they help restore SAM activity, improve detoxification, and strengthen mitochondrial function.
When methylation and energy metabolism are aligned, ATP production stabilizes, oxidative stress declines, and cognitive and behavioral symptoms often improve. In this way, methylation is not just a mood pathway—it’s the biochemical bridge between detoxification, energy, and the neurological balance seen in healthy brain function.
Oxidative Stress and Autism– The Missing Link
Every time mitochondria make ATP, they generate small amounts of reactive oxygen species (ROS)—tiny but reactive molecules that act like sparks from an engine.
Normally, antioxidants such as glutathione, catalase, and superoxide dismutase extinguish these sparks before they cause harm. But when mitochondria are already strained or methylation is impaired, the body’s antioxidant defenses falter. ROS accumulate, damaging membranes, enzymes, and DNA.
This oxidative stress in autism is widely observed and helps explain why many individuals experience rapid fatigue, sensitivity to light or sound, and difficulty handling infections or toxins.
How to Support Energy and Reduce Oxidative Stress
Restoring mitochondrial and methylation balance requires giving both systems the nutrients and environment they need to cooperate again.
1. Support mitochondrial energy production
-
Magnesium, CoQ10, L-carnitine, and alpha-lipoic acid (ALA) strengthen mitochondrial enzymes and improve ATP synthesis.
-
Adequate protein and healthy fats supply clean fuel for the energy cycle.
2. Restore methylation flow
-
SAMe, methionine, zinc, vitamin B6, and creatine provide the raw materials and cofactors for methylation.
-
Correcting these imbalances raises glutathione and helps mitochondria produce energy more cleanly.
3. Reduce oxidative and inflammatory load
-
A low-glycemic Mediterranean-style diet rich in vegetables, olive oil, eggs, garlic, and cruciferous plants provides antioxidants, sulfur, and minerals that feed detox pathways.
-
Avoiding processed foods and artificial additives lowers the burden on mitochondria and the liver.
Together, these steps create the conditions for better focus, calmer mood, and improved recovery from daily stressors.
Why Energy Equals Function
When methylation and mitochondria are synchronized, cells create energy efficiently and neutralize toxins before they cause harm.
This steady energy translates into better brain signaling, emotional resilience, and developmental progress. It’s not just a biochemical correction—it’s restoring the body’s ability to run its own repair systems.
The Takeaway
Mitochondrial dysfunction and undermethylation are two sides of the same coin. Weak methylation limits the antioxidants mitochondria need; struggling mitochondria increase oxidative stress that further slows methylation.
Addressing both is key to improving energy, detox, and behavior in autism. With the right nutritional and biochemical support, many individuals experience clearer thinking, steadier emotions, and a new level of vitality.


Elderly spouse cognition snd gait issues.
Also high iron sat, low homocysteine, low T3, high SHBG and assume elevated histamine
He has snps for HNMT, DAO enzyme , hepcidin deficiency and I believe one allele for CBS upregulation
He tested high for antibody intro is factor.
I tried low dose methylB12 very bad reaction.
Gets dehydrated so easily because of severe constpation needing laxative help
He reacts horribly to everything
I believe he is under methylated due to his characteristics
He has fast MAO and intermediate COMT
He reacts poorly to just about everything we try
He is down to 5’4 and 120 weight
He was a athlete professional cyclist
He always ate very healthy, exercised and never drank nor smoked anything
He was hit by car 4 years ago on bike so everything has intensified.
Prior to that cognition issues and high iron sat and ferritin
He donated blood but reacted poorly after besides decreasing his total iron intake
After listening to Levy probably not as good as we should have
So I just started giving creatine very low dose 500mg.
Thought about trying lactoferrin low dose also
Maybe doing the methylation panel ?
I’ve done so many lab studies trying to get answers and many different doctors and continue to spin my wheels trying to help this man.
Any suggestions?
He may react poorly to therapies because his methylation capacity is low. Methylation is necessary to metabolize compounds so they can be removed from the body. B12 is not a measure of need, though a methylation pathway panel can tell if he has low methylation from low methionine and low SAM or if he has high SAH, an “anti-methylator.” My suspicion is that he has high SAH. This compound is used to produce homocysteine, so if homocysteine is low, that gives a clue that either SAH is high, and not converting to homocysteine, or that the entire methylation pathway is subpar. That is also possible, since his weight is low, he has key genetic SNPS, and his muscles are trained for high creatine demand, which requires rigorous methylation and he probably does not eat much meat or methionine containing proteins. Constipation is another indicator that SAH is elevated. Creatine is a good start but that dose is insufficient. Otherwise, while the iron information is not clear, oxidative stress from iron and copper are a big cause of cognitive decline. With elevated SHBG and low T3 he would probably benefit from bioidentical hormone therapy – which can restore vitality. He needs a good workup.